Pharmaceutical Knowledge Graph for Explainable Research and Regulatory Decisions
d.AP connects fragmented data into a shared model of meaning that supports faster research insights, explainable AI, and defensible regulatory decisions.
- Connect research, clinical, and regulatory data
- Understand relationships across molecules, trials, and outcomes
- Enable explainable AI insights through Aluna
- Support faster, compliant decision-making across the drug lifecycle

Why Pharmaceutical Data Alone Doesn’t Deliver Research Insight
Pharmaceutical organizations generate vast amounts of data across:
- Research environments
- Clinical trials
- Regulatory submissions
- Manufacturing and quality systems
Yet critical insights often remain difficult to obtain.
Because while the data exists, the relationships between research findings, clinical outcomes, and regulatory requirements are rarely modeled explicitly.
Drug development is built on relationships. Most enterprise data platforms are not.
From Siloed to Semantic Pharmaceutical Data
Traditional pharmaceutical data platforms store or move data across systems.
d.AP focuses on modeling meaning and relationships.
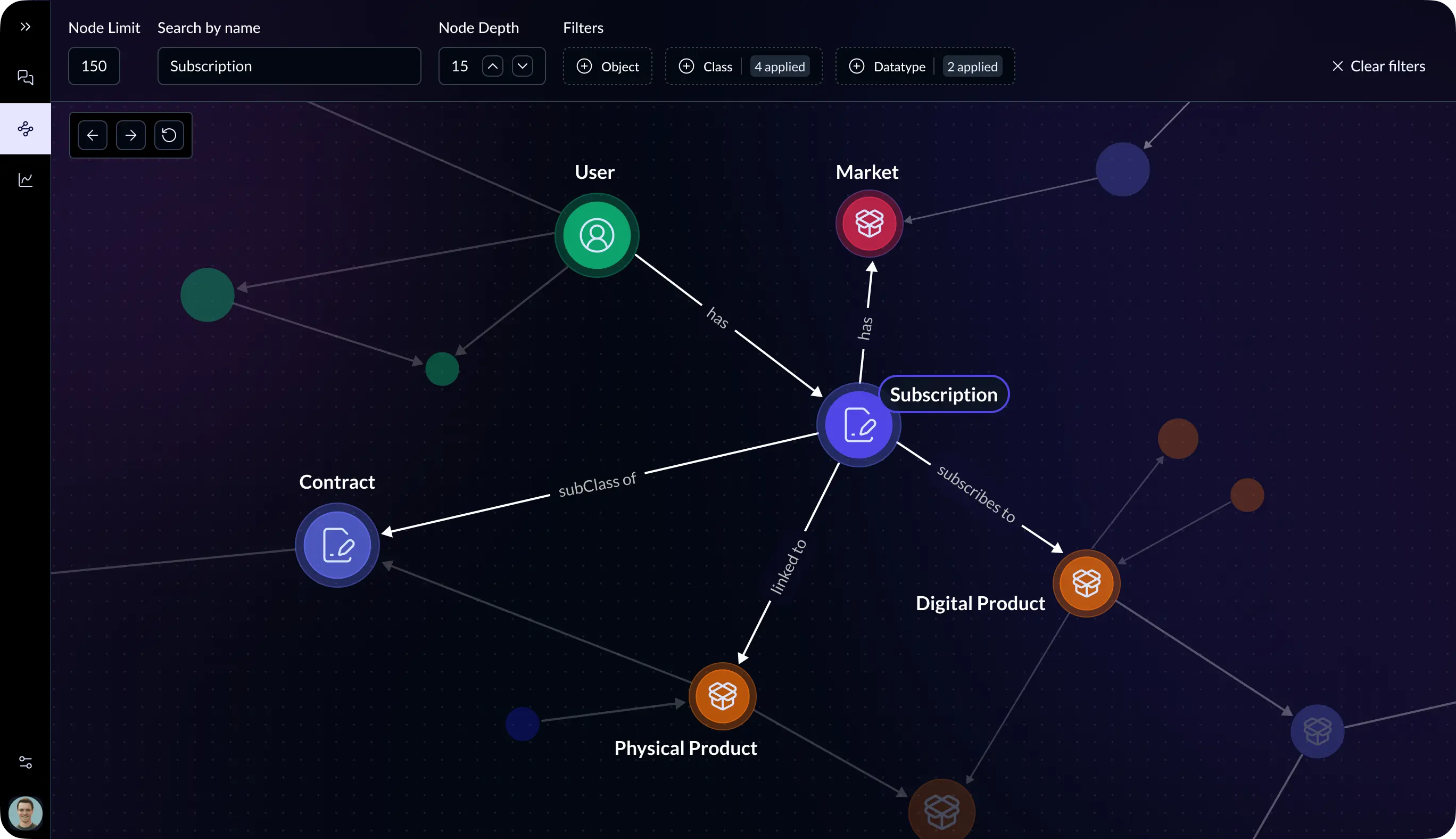
The platform builds an ontology-grounded Knowledge Graph representing:
- Molecules and compounds
- Clinical trials and patient outcomes
- Regulatory requirements
- Manufacturing and quality relationships
This shared model allows teams and AI systems to reason across the entire drug lifecycle.
Traditional Pharma Data Platforms
- Data fragmented across research systems
- Relationships inferred manually
- Insights recreated repeatedly
Pharma Knowledge Graph with d.AP
- Explicit scientific relationships
- Shared research context
- Reusable reasoning across teams
Step 1: Data Integration
Research, clinical, regulatory, and operational systems are connected into the Knowledge Graph.
Step 2: Ontology Modeling
Scientific entities, relationships, and constraints are modeled explicitly.
Step 3: Knowledge-Based Reasoning
Relationships and constraints enable reasoning across drug development processes.
Step 4: Explainable AI via Aluna
Teams ask questions in plain English and receive answers grounded in pharmaceutical knowledge.
Real results, real impact.
Accelerated Research Insight
Connect research findings, clinical data, and molecular knowledge to uncover relationships faster.
Explainable AI for Scientific Decisions
Aluna provides answers grounded in the pharmaceutical Knowledge Graph, ensuring transparency and regulatory confidence.
Improved Regulatory Traceability
Track relationships between data, evidence, and regulatory requirements across the drug lifecycle.
Questions d.AP Can Help Answer
Explainable AI for Pharmaceutical Decision Making
d.AP exposes the pharmaceutical Knowledge Graph through Aluna, allowing teams to ask questions in natural language and receive answers grounded in scientific knowledge.
Every answer returned through Aluna is:
- Derived from the Knowledge Graph
- Grounded in domain ontologies
- Traceable to relationships and constraints
This allows AI to support research and regulatory decisions without hallucination or guesswork.
Designed for Pharmaceutical Enterprise Architectures
d.AP integrates with existing research, clinical, and regulatory systems.
Deployment options include:
- EU-hosted SaaS (VPC)
- Customer-managed cloud environments
- Hybrid deployments
Most organizations begin with a focused domain such as research data or clinical trial knowledge.
Who d.AP is for
Frequently Asked Questions
We answer your questions in advance. We've missed something? Let us know.
Yes. d.AP is designed for environments where decisions must be transparent and traceable. Its ontology-driven Knowledge Graph makes relationships, assumptions, and evidence explicit, helping teams explain how conclusions are derived.
d.AP integrates with existing systems such as research databases, clinical data platforms, and regulatory systems. These systems remain the systems of record. d.AP connects their data into a Knowledge Graph so relationships across research, trials, and outcomes can be understood and reused.
Pharmaceutical data platforms typically store information but do not model the relationships between research findings, clinical outcomes, and regulatory evidence. d.AP creates a shared knowledge layer that connects these elements, allowing teams to reason across the entire drug development lifecycle.
d.AP provides the structured knowledge foundation required for trustworthy AI. Through Aluna, users can ask questions in plain English and receive answers derived from the Knowledge Graph. Because the answers are grounded in defined relationships and ontologies, they remain explainable and defensible.
Yes. d.AP is designed for large-scale environments where knowledge spans research, clinical trials, regulatory documentation, and operational systems. Its knowledge graph architecture allows relationships across these domains to be modeled and reasoned at enterprise scale.